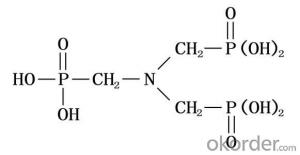
Hydroxypropyl Methycellulose
Hydroxypropyl Methycellulose Related Searches
Fiberglass Chemical Composition Industrial Urethane Alkyd Chemical Dop Scr420H Chemical Composition Methylcellulose AustraliaHot Searches
Hydroxypropyl Methycellulose Supplier & Manufacturer from China
Okorder.com is a professional Hydroxypropyl Methycellulose supplier & manufacturer, offers integrated one-stop services including real-time quoting and online cargo tracking. We are funded by CNBM Group, a Fortune 500 enterprise and the largest Hydroxypropyl Methycellulose firm in China.Hot Products
FAQ
- why is palladium/platinum a good catalyst?
- I'm guessing that the answer would be heterogeneous. Hydrogenation reactions use Pt and/or Pd metal as the catalyst. But these are always solids while the reaction is between H2 and either a gas or a liquid. Consequently the catalyst is in a different phase than the reactants, making them heterogeneous. I'd go with that one.
- What is the reaction of hydrogen peroxide to add manganese dioxide?
- There are bubbles produced
- Name one case in which catalyst poisoning is useful?
- alkynes can react with hydrogen in the presence of a catalyst such as palladium to form alkanes, if they catalyst is poisoned you can produce alkenes instead
- The quality and nature of the catalyst before and after the chemical reaction did not change this sentence wrong? Why did the "middle school student study" say it was wrong?
- The quality and nature of the catalyst are not changed before and after the chemical reaction ... 7604
- The last question asked no one answered this question.
- No other, is the test, so I put the pit out of the theory ... ...
- Chemical reaction in the presence of catalyst for the reaction are carried out a high life
- Some reactions require some of the catalyst that is not needed
- Is the chemical reaction rate constant related to the amount of catalyst used?
- The catalyst has a certain amount of suitable range, the general factory production of some substances (such as ammonia), the amount of catalyst used is limited, to achieve a value after no greater role. So the reaction rate constant is independent of the amount of catalyst used
- The "one-to-two change" of the catalyst is that the quality and chemical properties of the reactants are constant or the quality and chemical properties of the catalyst are constant?
- The quality and chemical properties of the catalyst are unchanged